10-Q: Quarterly report [Sections 13 or 15(d)]
Published on May 14, 2026
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
For
the quarterly period ended
Commission
file number:
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of | (I.R.S. Employer | |
| incorporation or organization) | Identification Number) |
(Address of principal executive offices, including Zip Code)
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer”, “accelerated filer”, “smaller reporting company”, and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ☐ | Accelerated filer ☐ |
| Smaller
reporting company | |
| Emerging
growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes
☐ No
As of May 12, 2026, the Company had shares of common stock issued and outstanding.
LIXTE BIOTECHNOLOGY HOLDINGS, INC.
AND SUBSIDIARY
TABLE OF CONTENTS
| 2 |
PART I - FINANCIAL INFORMATION
ITEM 1. CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
LIXTE BIOTECHNOLOGY HOLDINGS, INC.
AND SUBSIDIARY
CONDENSED CONSOLIDATED BALANCE SHEETS
| March 31, | December 31, | |||||||
| 2026 | 2025 | |||||||
| (Unaudited) | ||||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash | $ | $ | ||||||
| Prepaid insurance | ||||||||
| Other prepaid expenses | ||||||||
| Total current assets | ||||||||
| LiGHT proton therapy system equipment | ||||||||
| Operating lease right-of-use assets | ||||||||
| Total assets | $ | $ | ||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable and accrued expenses, including $ | $ | $ | ||||||
| Research and development contract liabilities | ||||||||
| Operating lease obligations, current | ||||||||
| Series B Convertible Preferred Stock 8% cumulative dividend payable | ||||||||
| Total current liabilities | ||||||||
| Operating lease obligations, long-term | ||||||||
| Total liabilities | ||||||||
| Commitments and Contingencies | ||||||||
| Stockholders’ Equity: | ||||||||
| Preferred stock, $ par value; shares authorized; | ||||||||
| Series A Convertible Preferred Stock, $ per share stated value – shares issued and outstanding at March 31, 2026 and December 31, 2025, respectively | ||||||||
| Series B Convertible Preferred Stock, $ per share stated value – shares issued and outstanding at March 31, 2026, and shares issued and outstanding at December 31, 2025 | ||||||||
| Common stock, $ par value; shares authorized; and shares issued and outstanding at March 31, 2026 and December 31, 2025, respectively | ||||||||
| Additional paid-in capital | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total Lixte Biotechnology stockholders’ equity | ||||||||
| Non-controlling interest | ||||||||
| Total stockholders’ equity | ||||||||
| Total liabilities and stockholders’ equity | $ | $ | ||||||
See accompanying notes to condensed consolidated financial statements.
| 3 |
LIXTE BIOTECHNOLOGY HOLDINGS, INC.
AND SUBSIDIARY
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS
(Unaudited)
| Three Months Ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Revenues, net | $ | $ | ||||||
| Costs and expenses: | ||||||||
| General and administrative costs | ||||||||
| Research and development costs | ||||||||
| Total costs and expenses | ||||||||
| Loss from operations | ( | ) | ( | ) | ||||
| Other income (expenses): | ||||||||
| Interest Income | ||||||||
| Interest Expense | ( | ) | ( | ) | ||||
| Foreign currency gain (loss) | ( | ) | ||||||
| Other income | ||||||||
| Net loss | $ | ( | ) | $ | ( | ) | ||
| Series B Convertible Preferred Stock 8% cumulative dividend | ( | ) | ||||||
| Non-controlling interest | $ | $ | ||||||
| Net loss attributable to common stockholders | $ | ( | ) | $ | ( | ) | ||
| Net loss per common share – basic and diluted | $ | ) | $ | ) | ||||
| Weighted average common shares outstanding – basic and diluted | ||||||||
See accompanying notes to condensed consolidated financial statements.
| 4 |
LIXTE BIOTECHNOLOGY HOLDINGS, INC.
AND SUBSIDIARY
CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(Unaudited)
| Series A Convertible Preferred Stock | Series B Convertible | Common Stock | Additional Paid in | Accumulated | Non-controlling | Total Stockholders’ | ||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | Capital | Deficit | interest | Equity | |||||||||||||||||||||||||||||||
| Balance, December 31, 2025 | $ | $ | $ | $ | $ | ( | ) | $ | $ | |||||||||||||||||||||||||||||||
| Exercise of pre-funded warrants | ( | ) | ||||||||||||||||||||||||||||||||||||||
| Fair value of shares issued for services | ||||||||||||||||||||||||||||||||||||||||
| Series B Convertible Preferred Stock 8% cumulative dividend | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||||||
| Conversion of Series B Convertible Preferred Stock and related dividend | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||||||
| Stock-based compensation | ||||||||||||||||||||||||||||||||||||||||
| Net loss | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||||
| Balance, March 31, 2026 | - | $ | - | $ | $ | $ | $ | ( | ) | $ | $ | |||||||||||||||||||||||||||||
Series A Convertible | Series B Convertible Preferred Stock | Common Stock | Additional Paid in | Accumulated | Non-controlling | Total Stockholders’ | ||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | Capital | Deficit | interest | Equity | |||||||||||||||||||||||||||||||
| Balance, December 31, 2024 | $ | $ | - | $ | $ | $ | ( | ) | $ | - | $ | |||||||||||||||||||||||||||||
| Proceeds from sale of securities registered direct offering, net of offering costs | - | - | - | - | ||||||||||||||||||||||||||||||||||||
| Stock options issued to settle accrued payable | - | - | - | - | - | |||||||||||||||||||||||||||||||||||
| Stock-based compensation | - | - | - | - | - | |||||||||||||||||||||||||||||||||||
| Net loss | - | - | - | - | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||
| Balance, March 31, 2025 | $ | $ | $ | $ | $ | $ | ( | ) | $ | $ | ||||||||||||||||||||||||||||||
See accompanying notes to condensed consolidated financial statements.
| 5 |
LIXTE BIOTECHNOLOGY HOLDINGS, INC.
AND SUBSIDIARY
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(Unaudited)
| Three Months Ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net loss | $ | ( | ) | $ | ( | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Non-cash operating lease cost | ||||||||
| Stock-based compensation expense | ||||||||
| Fair value of shares issued for services | ||||||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid insurance | ( | ) | ||||||
| Other prepaid expenses | ( | ) | ( | ) | ||||
| Accounts payable and accrued expenses | ( | ) | ||||||
| Research and development contract liabilities | ||||||||
| Operating lease liability | ||||||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||
| Capitalized costs for LiGHT equipment | ( | ) | ||||||
| Net cash used in investing activities | ( | ) | ||||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||
| Proceeds from sale of securities in registered direct offerings, net of offering costs | ||||||||
| Net cash provided by financing activities | ||||||||
| NET CHANGE IN CASH | ||||||||
| Net increase (decrease) | ( | ) | ||||||
| Cash - Beginning of period | ||||||||
| Cash - End of period | $ | $ | ||||||
| Supplemental disclosures of cash flow information: | ||||||||
| Cash paid for interest | $ | $ | ||||||
| Non-cash investing and financing activities: | ||||||||
| Settlement of accrued compensation to Board of Directors by issuance of stock options | $ | |||||||
| Exercise of pre-funded warrants | $ | $ | ||||||
| Conversion of Series B Convertible Preferred Stock into common stock and related dividend | $ | $ | ||||||
See accompanying notes to condensed consolidated financial statements.
| 6 |
LIXTE BIOTECHNOLOGY HOLDINGS, INC.
AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
Three Months Ended March 31, 2026 and 2025
1. Organization and Basis of Presentation
The unaudited condensed consolidated financial statements as of March 31, 2026, and for the three months ended March 31, 2026 and 2025, of Lixte Biotechnology Holdings, Inc. (a Delaware corporation), include the accounts of Lixte Biotechnology, Inc. (a wholly-owned Delaware corporation), and Liora Technologies Europe Ltd. (“Liora”, an 80% owned corporation organized under the laws of England and Wales) (collectively, the “Company”).
In the opinion of management of the Company, all adjustments, including normal recurring accruals, have been made that are necessary to present fairly the financial position of the Company as of March 31, 2026, and the results of its operations for the three months ended March 31, 2026, and its cash flows for the three months ended March 31, 2026. Operating results for the interim periods presented are not necessarily indicative of the results to be expected for a full fiscal year. The condensed consolidated balance sheet at December 31, 2025 has been derived from the Company’s audited consolidated financial statements at such date.
The condensed consolidated financial statements and related notes have been prepared pursuant to the rules and regulations of the Securities and Exchange Commission (“SEC”). Accordingly, certain information and footnote disclosures normally included in financial statements prepared in accordance with generally accepted accounting principles have been omitted pursuant to such rules and regulations. These condensed consolidated financial statements should be read in conjunction with the financial statements and other information included in the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2025, as filed with the SEC.
Business
The Company is a clinical-stage biopharmaceutical and proton cancer therapy company focused on identifying new targets for cancer drug development and developing and commercializing cancer therapies. The Company’s drug product pipeline is primarily focused on inhibitors of protein phosphatase 2A, which is used to enhance cytotoxic agents, radiation, immune checkpoint blockers and other cancer therapies. The Company believes that inhibitors of protein phosphatases have significant therapeutic potential for a broad range of cancers. The Company is focusing on the clinical development of a specific protein phosphatase inhibitor, referred to as LB-100, which has been shown to have clinical anti-cancer activity.
The Company is the majority shareholder of Liora Technologies Europe Ltd., which is pioneering the development of electronically controlled proton therapy systems for treating tumors in various types of cancers. Liora’s proprietary technology, known as LiGHT System (Linac for Image Guided Hadron Therapy), has significant advantages over currently available technologies for treating tumors with proton therapy. Liora is an excellent complement to the pharmaceutical side of the Company’s business and ongoing clinical trials with LB-100 for Ovarian Clear Cell Carcinoma and Metastatic Colon Cancer.
The Company’s activities are subject to significant risks and uncertainties, including the need for additional capital. The Company has not yet commenced any revenue-generating operations, does not have positive cash flows from operations, relies on stock-based compensation for a substantial portion of employee and consultant compensation, and is dependent on periodic infusions of equity capital to fund its operating requirements.
| 7 |
Going Concern
For
the three months ended March 31, 2026, the Company incurred a net loss of $
The
Company is currently engaged in early-stage clinical trials for its lead product candidate, LB-100. These activities require substantial
research, development, regulatory, and clinical expenditures, and the Company does not expect to generate sustainable operating revenues
for several years, if ever. At March 31, 2026, the Company’s remaining contractual commitments pursuant to clinical trial agreements
and clinical trial monitoring agreements aggregated approximately $
In
addition, the Company expects to incur approximately $
Management is actively evaluating and pursuing additional financing alternatives, including equity and debt financings and potential strategic transactions. However, there can be no assurance that additional funding will be available on acceptable terms, in sufficient amounts, or at all. If the Company is unable to obtain the necessary funding, it may be required to delay, scale back, or eliminate its clinical development programs; curtail expenditures related to the LiGHT system; or pursue strategic alternatives, including potential asset sales or the cessation of operations.
Nasdaq Compliance
The Company’s common stock is traded on the Nasdaq Capital Market under the symbol “LIXT”.
On
August 23, 2024, the Company received written notification from the Listing Qualifications Department (the “Staff”) of the
Nasdaq Stock Market LLC (“Nasdaq”) that the Company was not in compliance with the minimum stockholders’ equity requirement
of $
The Company remains subject to a Panel Monitor under Nasdaq Listing Rule 5815(d)(4)(B) through July 15, 2026. During this period, any future deficiency in stockholders’ equity would require the Company to request a hearing before the Panel rather than submit a new compliance plan.
| 8 |
2. Summary of Significant Accounting Policies
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities at the date of the financial statements and the reported amounts of expenses during the reporting period. Actual results could differ from those estimates. Significant estimates include those related to assumptions used in the calculation of accruals for clinical trial costs and other potential liabilities, and valuing equity instruments issued for services.
Asset Acquisitions
The Company assesses whether an acquisition is a business combination or an asset acquisition. If substantially all of the gross assets acquired are concentrated in a single asset or group of similar assets, then the acquisition is accounted for as an asset acquisition, where the purchase consideration is allocated on a relative fair value basis to the assets acquired. An asset acquisition does not result in the recognition of goodwill and transaction costs are capitalized as part of the cost of the asset or group of assets acquired. The Company uses its best estimates and assumptions to assign fair value to the tangible and intangible assets acquired and liabilities assumed at the acquisition date. The acquisitions costs are allocated to the assets acquired on a relative fair value basis.
Property and Equipment
The Company property and equipment consists of Liora’s LiGHT system equipment. Property and equipment are recorded at cost. The LiGHT machine requires recommissioning and updates and is not yet ready for its intended use. Accordingly, it is treated as an asset under construction, and depreciation will not begin until the asset is placed into service.
Long – Lived Assets
Long-lived assets, which include property, plant and equipment and operating lease right-of-use assets, are reviewed for impairment whenever events or changes in circumstances indicate the carrying amount of an asset may not be recoverable.
Recoverability of long-lived assets to be held and used is measured by comparing the carrying amount of an asset to the estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated undiscounted future cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the assets. Fair value is generally determined using the asset’s expected future discounted cash flows or market value, if readily determinable.
| 9 |
The
Company reviews long-lived assets for impairment whenever events or changes in circumstances indicate that the asset’s carrying
amount may not be recoverable. In conducting its long-lived asset impairment analyses, the Company groups assets and liabilities at the
lowest level for which identifiable cash flows are largely independent of the cash flows of other assets and liabilities and evaluates
the asset group against the sum of the undiscounted future cash flows. If the undiscounted cash flows do not indicate the carrying amount
of the asset is recoverable, an impairment charge is measured as the amount by which the carrying amount of the asset group asset group
exceeds its fair value based on discounted cash flow analysis or appraisals. There was
Research and Development
Research and development costs are charged to expense as incurred. The costs of equipment that are acquired or constructed for research and development activities, and have alternative future uses, are classified as property and equipment and depreciated over their estimated useful lives. Research and development costs consist primarily of fees paid to consultants and contractors, and other expenses relating to the negotiation, design, development, conduct and management of clinical trials with respect to the Company’s clinical compound and product candidate. Research and development costs also include the costs to manufacture compounds used in research and clinical trials, which are charged to operations as incurred. The Company’s inventory of LB-100 for clinical use has been manufactured separately in the United States and in the European Union in accordance with the laws and regulations of such jurisdictions.
Research and development costs are generally charged to operations ratably over the life of the underlying contracts, unless the achievement of milestones, the completion of contracted work, the termination of an agreement, or other information indicates that a different expensing schedule is more appropriate. However, payments for research and development costs that are contractually defined as non-refundable are charged to operations as incurred.
Obligations incurred with respect to mandatory scheduled payments under agreements with milestone provisions are recognized as charges to research and development costs in the Company’s consolidated statement of operations based on the achievement of such milestones, as specified in the respective agreement. Obligations incurred with respect to mandatory scheduled payments under agreements without milestone provisions are accounted for when due, are recognized ratably over the appropriate period, as specified in the respective agreement, and are recorded as liabilities in the Company’s consolidated balance sheet, with a corresponding charge to research and development costs in the Company’s consolidated statement of operations.
Payments made pursuant to contracts are initially recorded as advances on research and development contract services in the Company’s consolidated balance sheet and are then charged to research and development costs in the Company’s consolidated statement of operations as those contract services are performed. Expenses incurred under contracts in excess of amounts advanced are recorded as research and development contract liabilities in the Company’s consolidated balance sheet, with a corresponding charge to research and development costs in the Company’s consolidated statement of operations. The Company reviews the status of its various clinical trial and research and development contracts on a quarterly basis.
Patent, Licensing, Legal and Filing Fees and Costs
Due
to the significant uncertainty associated with the successful development of commercially viable products based on the Company’s
research efforts and related patent applications, all patent, licensing, legal and filing fees and costs related to the development
and protection of the Company’s intellectual property are charged to operations as incurred. Patent and licensing legal and filing
fees and costs were $
Concentration of Risk
The
Company periodically contracts with vendors and consultants to provide services related to the Company’s operations. Charges incurred
for these services can be for a specific period (typically one year) or for a specific project or task. Costs and expenses incurred that
represented
General
and administrative costs for the three months ended March 31, 2026 and 2025 include charges from legal firms and other vendors for general
licensing and patent prosecution costs relating to the Company’s intellectual properties representing
Research
and development costs for the three months ended March 31, 2026 include charges from two major vendors representing
Research
and development costs for the three months ended March 31, 2025 include charges from five vendors and consultants representing
| 10 |
The Company periodically issues common stock and stock options to officers, directors, employees, contractors and consultants for services rendered. Options vest and expire according to terms established at the issuance date of each grant. Stock grants, which are generally time vested, are measured at the grant date fair value and charged to operations ratably over the vesting period.
The Company accounts for stock-based payments to officers, directors, employees, contractors, and consultants by measuring the cost of services received in exchange for equity awards utilizing the grant date fair value of the awards, with the cost recognized as compensation expense on the straight-line basis in the Company’s financial statements over the vesting period of the awards. Recognition of compensation expense for non-employees is in the same period and manner as if the Company had paid cash for the services.
The fair value of stock options granted as stock-based compensation is determined utilizing the Black-Scholes option-pricing model, and is affected by several variables, the most significant of which are the expected life of the stock option, the exercise price of the stock option as compared to the fair market value of the common stock on the grant date, and the estimated volatility of the common stock. Unless sufficient historical exercise data is available, the expected life of the stock option is calculated as the mid-point between the vesting period and the contractual term (the “simplified method”). The estimated volatility is based on the historical volatility of the Company’s common stock, calculated utilizing a look-back period approximately equal to the contractual life of the stock option being granted. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant. The fair market value of the common stock is determined by reference to the quoted market price of the Company’s common stock on the grant date. The expected dividend yield is based on the Company’s expectation of dividend payouts and is assumed to be zero.
The Company recognizes the fair value of stock-based compensation awards in general and administrative costs and in research and development costs, as appropriate, in the Company’s consolidated statements of operations. The Company issues new shares of common stock to satisfy stock option exercises.
Warrants
The Company accounts for warrants as either equity-classified or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance in Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 480, Distinguishing Liabilities from Equity (“ASC 480”), and ASC 815, Derivatives and Hedging (“ASC 815”). The assessment considers whether the warrants are freestanding financial instruments pursuant to ASC 480, meet the definition of a liability pursuant to ASC 480, and whether the warrants meet all of the requirements for equity classification under ASC 815, including whether the warrants are indexed to the Company’s own common stock and whether the warrant holders could potentially require “net cash settlement” in a circumstance outside of the Company’s control, among other conditions for equity classification. The Company has determined that the warrants issued in the February 2025 equity financing, and the July 2025 equity financings (see Note 5) meet the requirements for equity classification. For issued or modified warrants that meet all of the criteria for equity classification, the warrants are required to be recorded as a component of additional paid-in capital at the time of issuance. For issued or modified warrants that do not meet all of the criteria for equity classification, the warrants are required to be liability-classified and recorded at their initial fair value on the date of issuance and remeasured at fair value at each reporting date. At March 31, 2026 and December 31, 2025, the Company did not have any liability-classified warrants.
Segment Information
The Company’s Chief Executive Officer is the Company’s Chief Operating Decision Maker (“CODM”) and evaluates performance and makes operating decisions about allocating resources based on internal financial data presented on a consolidated basis. Because the CODM evaluates financial performance on a consolidated basis, the Company has determined that it operates in a single reportable segment, which consists of the development of cancer treatments. The CODM uses consolidated net income (loss) as the sole measure of segment profit or loss.
| 11 |
The Company’s computation of earnings (loss) per share (“EPS”) includes basic and diluted EPS. Basic EPS is measured as the income (loss) attributable to common stockholders divided by the weighted average common shares outstanding for the period. Diluted EPS is similar to basic EPS but presents the dilutive effect on a per share basis of potential common shares (e.g., preferred shares, warrants and stock options) as if they had been converted at the beginning of the respective periods presented, or issuance date, if later. Potential common shares that have an anti-dilutive effect (i.e., those that increase income per share or decrease loss per share) are excluded from the calculation of diluted EPS.
Loss per common share is computed by dividing net loss by the weighted average number of common shares outstanding during the respective periods. The weighted average number of common shares outstanding utilized for determining basic net loss per common share for the three months ended March 31, 2026 includes all pre-funded warrants sold that have not been exercised as of March 31, 2026. Basic and diluted loss per common share was the same for all periods presented because all preferred shares, warrants (excluding pre-funded warrants) and stock options outstanding were anti-dilutive.
| March 31, | ||||||||
| 2026 | 2025 | |||||||
| Series A Convertible Preferred Stock | ||||||||
| Common stock warrants (excluding pre-funded warrants) | ||||||||
| Common stock options | ||||||||
| Total | ||||||||
Foreign Currency Translation
The consolidated financial statements are presented in the United States dollar, which is the functional and reporting currency of the Company.
The Company periodically incurs a cost or expense in a foreign jurisdiction denominated in a local currency. The Company purchases the required foreign currency to pay such cost or expense on an as-needed basis. Such cost or expense is converted into United States dollars for financial statement purposes based on the foreign currency conversion rate in effect on the transaction date. The Company purchases the requisite foreign currency to pay such cost or expense on an as-needed basis. Any gain or loss resulting from the purchase of the foreign currency is included as realized foreign currency gain (loss) in the consolidated statement of operations.
During
the three months ended March 31, 2026 and 2025, the Company incurred various costs and expenses denominated in Euros, which were converted
into United States dollars at the average rate of
| 12 |
Fair Value of Financial Instruments
The authoritative guidance with respect to fair value established a fair value hierarchy that prioritizes the inputs to valuation techniques used to measure fair value into three levels and requires that assets and liabilities carried at fair value be classified and disclosed in one of three categories, as presented below. Disclosure as to transfers in and out of Levels 1 and 2, and activity in Level 3 fair value measurements, is also required.
Level 1. Observable inputs such as quoted prices in active markets for an identical asset or liability that the Company has the ability to access as of the measurement date. Financial assets and liabilities utilizing Level 1 inputs include active-exchange traded securities and exchange-based derivatives.
Level 2. Inputs, other than quoted prices included within Level 1, which are directly observable for the asset or liability or indirectly observable through corroboration with observable market data. Financial assets and liabilities utilizing Level 2 inputs include fixed income securities, non-exchange-based derivatives, mutual funds, and fair-value hedges.
Level 3. Unobservable inputs in which there is little or no market data for the asset or liability which requires the reporting entity to develop its own assumptions. Financial assets and liabilities utilizing Level 3 inputs include infrequently traded non-exchange-based derivatives and commingled investment funds and are measured using present value pricing models.
The Company determines the level in the fair value hierarchy within which each fair value measurement falls in its entirety, based on the lowest level input that is significant to the fair value measurement in its entirety. In determining the appropriate levels, the Company performs an analysis of the assets and liabilities at each reporting period end.
The carrying value of financial instruments, which consists of accounts payable and accrued expenses, is considered to be representative of their respective fair values due to the short-term nature of those instruments. The carrying value of digital assets is based on quoted prices in active markets (Level 1 inputs).
Recent Accounting Pronouncements
In November 2024, the Financial Accounting Standards Board (the “FASB”) issued Accounting Standards Update (“ASU”) 2024-03 “Income Statement – Reporting Comprehensive Income – Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses.” This ASU requires public business entities to disclose, for interim and annual reporting periods, additional information about certain income statement expense categories. The requirements are effective for fiscal years beginning after December 15, 2026, and for interim periods beginning after December 15, 2027. Entities are permitted to apply either the prospective or retrospective transition methods. The Company is in the process of evaluating the adoption of this ASU to determine its impact on the Company’s disclosures.
In December 2025, the FASB issued ASU No. 2025-11, Interim Reporting (Topic 270): Narrow-Scope Improvements, which includes amendments to clarify interim reporting requirements and applicability of Topic 270 and codifies a principle requiring disclosure of material events and changes since the most recent annual reporting period. This guidance is effective for the Company for interim periods within fiscal years beginning after December 15, 2027, with early adoption permitted. The Company is in the process of evaluating the impact of adoption of this ASU on its Condensed Consolidated Financial Statements.
| 13 |
The Company’s management has evaluated all other recently issued, but not yet effective, accounting standards and guidance that have been issued or proposed by the FASB or other standards-setting bodies through the filing date of these financial statements and does not believe the future adoption of any such pronouncements will have a material effect on the Company’s financial position and results of operations.
3. Segment Information
The Company’s chief operating decision maker (“CODM”) has been identified as the Company’s Chief Executive Officer (“CEO”). The Company’s CODM evaluates performance and makes operating decisions about allocating resources based on financial data presented on a consolidated basis. Because the CODM evaluates financial performance on a consolidated basis, the Company has determined that it currently has a single operating segment which is comprised of the consolidated financial results of the Company.
The following table presents the significant segment expenses (10% or greater) and other segment items regularly reviewed by the Company’s CODM and included in research and development costs for the three months and March 31, 2026 and 2025.
| Three Months Ended | ||||||||
| March 31, | ||||||||
| 2026 | 2025 | |||||||
| Clinical and related oversight costs | $ | $ | ||||||
| Preclinical research focused on development of additional novel anti-cancer compounds | ||||||||
| Regulatory service costs | ||||||||
| Total research and development costs | $ | $ | ||||||
| 14 |
The following table presents a summary of research and development costs for the three months ended March 31, 2026 and 2025 based on the respective geographical regions where such costs were incurred.
| Three Months Ended | ||||||||
| March 31, | ||||||||
| 2026 | 2025 | |||||||
| United States | $ | $ | ||||||
| Spain | ||||||||
| Total | $ | $ | ||||||
The following table presents the significant segment expenses (10% or greater) and other segment items regularly reviewed by the Company’s CODM and included in general and administrative costs for the three months ended March 31, 2026 and 2025.
| Three Months Ended | ||||||||
| March 31, | ||||||||
| 2026 | 2025 | |||||||
| Compensation: | ||||||||
| Cash based compensation | $ | $ | ||||||
| Stock-based compensation | ||||||||
| Patent and licensing legal and filing fees and costs | ||||||||
| Marketing and public relations | ||||||||
| Accounting and bookkepping services | ||||||||
| Audit and legal | ||||||||
| Consulting fees | ||||||||
| Insurance expense | ||||||||
| Other costs and expenses, net | ||||||||
| Total general and administrative costs | $ | $ | ||||||
4. Acquisition of Liora Technologies Europe Ltd.
On
November 21, 2025, the Company entered into a share exchange agreement to acquire Liora from Orbit Capital. The acquisition was
executed through multiple agreements, including (i) an initial share exchange agreement dated November 21, 2025 (the “Original
SEA”); (ii) a subsequent share exchange agreement dated December 30, 2025 (the “Post-Closing SEA”); and (iii) an
amended and restated agreement dated March 6, 2026 (the “A&R Agreement”). The Post-Closing SEA finalized the
structure such that Lixte owned
| 15 |
The Company evaluated whether the acquisition met the definition of a business. Management determined that substantially all of the fair value of the assets acquired is concentrated in the LiGHT proton therapy system equipment, and that the acquisition of Liora did not include substantive processes or an organized workforce. Accordingly, the acquisition does not meet the definition of a business and is accounted for as an asset acquisition.
The following table summarizes the fair value of the purchase consideration and the fair value of tangible assets and assumed liabilities of Liora on the date of acquisition:
| Total consideration transferred and implied fair value of acquired assets | ||||
| Cash | $ | |||
| Digital assets (10.5925 Bitcoin BTC) | ||||
| Digital assets (300.699 Ether ETH) | ||||
| Common stock (700,000 shares of common stock at $ per share) | ||||
| Total consideration transferred for | ||||
| Noncontrolling interest ( | ||||
| The implied total value of the acquired asset ( | ||||
| Capitalized transaction costs | ||||
| Total acquisition costs | $ | |||
| Liora’s identifiable assets acquired and liabilities assumed | ||||
| LIGHT proton therapy system equipment | $ | |||
In January 2026, the Company capitalized an additional
$
The
LiGHT system is presented as a consolidated asset. The
Contingent consideration
Liora’s
purchase of the LiGHT system from AVO included deferred milestone-based payments of $
Lease accounting
The LiGHT machine is located in a leased facility. After the acquisition of the LiGHT machine from AVO, Liora entered into a two year operating lease with the Daresbury Laboratory site on November 17, 2025. The Company assumed the lease obligations and is accounting for the lease under ASC 842, including recognition of a right-of-use asset and lease liability (see Note 5). Currently, the Company expects that the LiGHT machine will continue to be housed at this location on a long-term basis.
| 16 |
5. Leases
On November 21, 2025, the Company assumed, as part
of the Liora acquisition, a two-year lease agreement with United Kingdom Research and Innovation for the rental of the premises at UKRI
Daresbury that houses the acquired LiGHT system equipment. The lease was classified as an operating lease and has a quarterly base rent
of GBP
Supplemental cash flow information related to operating leases is as follows:
Three months ended March 31, 2026 | ||||
| Cash payments for operating leases | ||||
| Weighted-average remaining lease term (in years) | ||||
| Weighted-average discount rate | % | |||
The Company’s operating lease right of use asset
was $
The Company’s operating lease liability balance
was $
During the three months ended March 31, 2026 and 2025,
lease costs totaled approximately $
As of March 31, 2026, the weighted average remaining
lease terms for operating lease is
Maturities of the Company’s operating lease liabilities are as follows as of March 31, 2026:
As of March 31, 2026 | ||||
| 2026 (remaining) | $ | |||
| 2027 | ||||
| Thereafter | ||||
| Total | ||||
| Less: Imputed interest | ( | ) | ||
| Total operating lease liability | $ | |||
6. Stockholders’ Equity
Preferred Stock
The Company is authorized to issue a total of shares of preferred stock, par value $ per share. On March 17, 2015, the Company filed a Certificate of Designations, Preferences, Rights and Limitations of its Series A Convertible Preferred Stock with the Delaware Secretary of State to amend the Company’s certificate of incorporation. The Company has designated a total of shares as Series A Convertible Preferred Stock, which are non-voting.
On October 21, 2025, the Company filed a Certificate of Elimination of Certificate of Designations of Series A Convertible Preferred Stock with the Delaware Secretary of State to eliminate the shares of Preferred Stock associated with the Series A Convertible Preferred Stock classification.
| 17 |
On
July 1, 2025, the Company filed amendment to Certificate of Incorporation by filing Certificate of Designations, Preferences, Rights
and Limitations of its Series B Convertible Preferred Stock with the Delaware Secretary of State. The Company has designated a total
of shares as Series B Convertible Preferred Stock with a stated value of $ per share. Each Preferred Share is convertible
into one share of Common Stock, subject to standard adjustments such as stock splits and stock dividends. The Preferred Shares are non-voting,
except that certain actions of the Company may not be taken except upon approval of holders who own a majority in stated value of the
Preferred Shares. The Preferred Shares bear an
As of March 31, 2026 and December 31, 2025 the Company had 10,000,000 and shares respectively, of undesignated preferred stock, which may be issued with such rights and powers as the Board of Directors may designate.
Common Stock
The Company is authorized to issue a total of shares of common stock, par value $ per share. As of March 31, 2026 and December 31, 2025, the Company had shares and shares, respectively, of common stock issued and outstanding.
2026 common stock transactions
Shares issued for services
On January 6, 2026, the Company entered into a consulting agreement with Pillow Hog Ventures, Inc (“PHVC”) for marketing and strategic consulting services. The agreement is for a term of six months ending June 30, 2026. The agreement provides for the payment to PHVC of shares of the Company’s common stock.
Exercise of Preferred Series B Stock
During January and February 2026, holders of the Company’s Preferred Series B stock converted an aggregate
of preferred shares, together with accrued cumulative dividends of $
Exercise of Pre-Funded Warrants
During the three months ended March 31, 2026, warrant holders exercised pre-funded warrants resulting in the issuance of shares of the Company’s common stock.
2025 common stock transactions
February 2025 equity offering
Effective
February 13, 2025, the Company closed a registered direct offering with certain investors which resulted in gross proceeds of $
All warrants issued in the February 2025 equity offering include customary anti-dilution adjustments and a “fundamental transaction” provision. If a qualifying fundamental transaction within the Company’s control is consummated, holders may elect cash settlement equal to the Black-Scholes value. For fundamental transactions outside the Company’s control, holders are entitled to receive the same consideration as common shareholders. The warrants are classified in permanent equity. Any future cash settlements will be accounted for as equity distributions upon occurrence of the related fundamental transaction.
| 18 |
Common Stock Warrants
A summary of common stock warrant activity, including warrants to purchase common stock that were issued in conjunction with the Company’s public offerings, but excluding pre-funded warrants, is presented below.
| Number of Shares | Weighted Average Exercise Price | Weighted Average Remaining Contractual Life (in Years) | ||||||||||
| Warrants outstanding at December 31, 2025 | ||||||||||||
| Issued | ||||||||||||
| Exercised | ||||||||||||
| Expired | ( | ) | ||||||||||
| Warrants outstanding at March 31, 2026 | ||||||||||||
At March 31, 2026, the outstanding warrants are exercisable at the following prices per common share:
| Exercise Price | Warrants Outstanding (Shares) | |||||
| $ | ||||||
During
the periods presented, the Company issued pre-funded warrants, each of which is exercisable immediately upon issuance at a de minimis
exercise price of $
Due to their economic characteristics, pre-funded warrants function as share-like instruments, rather than traditional warrants with a substantive exercise price or term. Therefore, including them together with standard warrants in the warrant rollforward would significantly distort both the weighted-average exercise price and the weighted-average remaining contractual life, rendering those disclosures not meaningful. As a result, the Company presents pre-funded warrants separately from standard common stock warrants in the tables below. The pre-funded warrants are excluded from weighted-average exercise price and remaining life due to their de-minimis strike price and share-like characteristics.
The following table presents a summary of activities related to pre-funded warrants.
| Number of Shares | ||||
| Pre-funded warrants outstanding at December 31, 2025 | ||||
| Issued | ||||
| Exercised | ( | ) | ||
| Expired | ||||
| Pre-funded warrants outstanding at March 31, 2026 | ||||
The following table presents a summary of total number of common stock warrants and pre-funded warrants.
| Warrant Type | 3/31/2026 | |||
| Common stock warrants | ||||
| Pre-funded warrants | ||||
| Total warrants outstanding | ||||
| 19 |
7. Related Party Transactions
Related party transactions include transactions with the Company’s officers, directors and affiliates.
Employment Agreements with Officers
Effective
June 16, 2025, the Company entered into an employment agreement with Geordan Pursglove pursuant to which Mr. Pursglove was appointed
as the Company’s Chief Executive Officer and Chairman of the Board of Directors for a term of three years, subject to automatic
termination if the Company did not complete a successful financing that would enable it to maintain its listing on the Nasdaq Capital
Market by July 3, 2025, which was accomplished on July 2, 2025. Under the employment agreement, Mr. Pursglove will receive an annual
salary of $
Effective
September 1, 2025, the Company entered into an employment agreement with Peter Stazzone to act as the Company’s Chief Financial
Officer, for a term of one year, with an annual salary of $
In
2023, the Company entered into an employment agreement with Bas van der Baan to act as the Company’s President, Chief Executive
Officer. Effective October 6, 2023, Mr. van der Baan was appointed as Chairman of the Board of Directors. Effective June 16, 2025, the
employment agreement was amended to provide that Mr. van der Baan will serve as President and Chief Scientific Officer of the Company.
Effective September 1, 2025, Mr. van der Bann resigned as President, but remained as the Company’s Chief Scientific Officer. The
term of the employment agreement is for three years and is automatically renewable for additional one-year periods. During the three
months ended March 31, 2026 and 2025, the Company paid $
Former officers
In
2020, the Company entered into an employment agreement with Robert N. Weingarten to act as the Company’s Vice President and Chief
Financial Officer. Mr. Weingarten resigned from the Company on September 1, 2025. During the three months ended March 31, 2025, the Company
paid $
In
2024, the Company entered into a consulting agreement with Dr. Jan H.M. Schellens, M.D., Ph.D. the Company engaged Dr. Schellens as a
consultant, and, effective August 1, 2024, as the Company’s Chief Medical Officer. The Company pays Dr. Schellens an annual compensation
of Euros (approximately $ as of December 31, 2025). Effective as of July 31, 2025, the Company agreed to accept the resignation
of Dr. Schellens. During the three months ended March 31, 2025, the Company paid $
In
2022, René Bernards was appointed to the Company’s Board of Directors as an independent director. and would receive annual
compensation for his services on the Board only in the form of cash, in lieu of the annual June 30 grant of stock options as provided
to the Company’s other non-officer directors. During the three months ended March 31, 2025, the Company recorded charges of $
Compensatory Arrangements for Members of the Board of Directors
Total cash compensation paid to non-officer directors was $ and $, respectively, for the three months ended March 31, 2026 and 2025.
Stock-based compensation granted to members of the Company’s Board of Directors, officers and affiliates is described at Note 8.
| 20 |
The Company periodically issues common stock and stock options as incentive compensation to directors and as compensation for the services of employees, contractors, and consultants of the Company.
Stock Options Issued, Vested and Cancelled
The Company periodically issues stock options as incentive compensation to directors and as compensation for the services of employees, contractors, and consultants of the Company.
As of March 31, 2026, unexpired stock options for shares were issued and outstanding under the 2020 Plan and shares were available for issuance under the 2020 Plan.
During the three months ended March 31, 2026, the Company did not grant any stock options.
On
January 20, 2025, the Company granted stock options to four non-officer directors of the Company to purchase shares of the Company’s
common stock, exercisable for a period of at an exercise price of $ per share (the closing market price on the grant date).
The grant date fair value of the stock options determined pursuant to the Black-Scholes option-pricing model was determined to be $
On
March 31, 2025, the Company granted to four non-officer directors of the Company a total of stock options to purchase shares of
the Company’s common stock, exercisable for a period of at an exercise price of $ per share (the closing market
price on the grant date). The stock options are exercisable for a period of five years and were immediately vested. The grant date fair
value of the stock options determined pursuant to the Black-Scholes option-pricing model was determined to be $
The total fair value of options that vested during the three months ended March 31, 2026 and 2025, was $ and $ respectively.
The fair value of a stock option award is calculated on the grant date using the Black-Scholes option-pricing model. The risk-free interest rate is based on the U.S. Treasury yield curve in effect as of the grant date. The expected dividend yield assumption is based on the Company’s expectation of dividend payouts and is assumed to be zero. The estimated volatility is based on the historical volatility of the Company’s common stock, calculated utilizing a look-back period approximately equal to the contractual life of the stock option being granted. Unless sufficient historical exercise data is available, the expected life of the stock option is calculated as the mid-point between the vesting period and the contractual term (the “simplified method”). The fair market value of the common stock is determined by reference to the quoted market price of the common stock on the grant date.
For stock options requiring an assessment of value during the three months ended March 31, 2025, the fair value of each stock option award was estimated using the Black-Scholes option-pricing model with the following assumptions:
| Risk-free interest rate | % | |||
| Expected dividend yield | % | |||
| Expected volatility | % | |||
| Expected life | years |
| 21 |
| Three Months Ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Officers and directors of the Company | $ | $ | ||||||
| Non-related parties | ||||||||
| Stock-based compensation costs | $ | $ | ||||||
A summary of stock option activity during the three months ended March 31, 2026 is as follows:
| Number of Shares | Weighted Average Exercise Price | Weighted Average Remaining Contractual Life (in Years) | ||||||||||
| Stock options outstanding at December 31, 2025 | $ | |||||||||||
| Granted | ||||||||||||
| Exercised | ||||||||||||
| Expired | ( | ) | ||||||||||
| Stock options outstanding at March 31, 2026 | ||||||||||||
| Stock options exercisable at December 31, 2025 | ||||||||||||
| Stock options exercisable at March 31, 2026 | $ | |||||||||||
As
of March 31, 2026, stock options were vested and exercisable. Total deferred compensation expense for the outstanding value
of unvested stock options was approximately $
| Exercise Prices | Options Outstanding (Shares) |
Options Exercisable (Shares) |
||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | |
|||||||||
| $ | ||||||||||
| $ | ||||||||||
| $ | ||||||||||
Based on the closing fair market value of $ per common share on March 31, 2026, the intrinsic value attributed to exercisable but unexercised common stock options was approximately $ at March 31, 2026.
Outstanding stock options to acquire shares of the Company’s common stock had not vested at March 31, 2026.
| 22 |
9. Commitments and Contingencies
Legal Claims
The Company may be subject to legal claims and actions from time to time as part of its business activities. As of March 31, 2026 and December 31, 2025, the Company was not subject to any threatened or pending lawsuits, legal claims or legal proceedings.
Principal Commitments
Clinical Trial Agreements
At
March 31, 2026, the Company’s remaining financial contractual commitments pursuant to clinical trial agreements and clinical trial
monitoring agreements not yet incurred, as described below, aggregated $
The following is a summary of the Company’s ongoing active contractual clinical trials described below as of March 31, 2026:
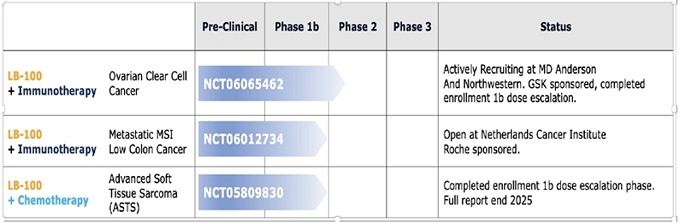
| Description of Clinical Trial | Institution | Start Date | Projected End Date | Number of Patients in Trial | Study Objective | Clinical Update | Expected Date of Preliminary Efficacy Signal | NCT No. | Remaining Financial Contractual Commitment | |||||||||||||
| Netherlands Cancer Institute (NKI) | Determine RP2D with atezolizumab | First patient entered August 2024, in total two patients entered | NCT06012734 | (1 | ) | |||||||||||||||||
| GEIS | Determine MTD and RP2D | Fourteen patients entered |
NCT05809830 | $ | ||||||||||||||||||
| GEIS | Determine efficacy: PFS | Clinical trial not yet begun (subject to completion of Phase 1b GEIS clinical trial) | NCT05809830 | $ | (1 | ) | ||||||||||||||||
| MD Anderson | Determine the OS of patients with recurrent ovarian clear cell carcinoma | Twenty one patients entered | NCT06065462 | (1 | ) | |||||||||||||||||
| Total | $ | |||||||||||||||||||||
| (1) |
| 23 |
Netherlands Cancer Institute. In June, 2024, the Company entered into a Clinical Trial Agreement with the Netherlands Cancer Institute (“NKI”) to conduct a Phase 1b clinical trial of the Company’s protein phosphatase inhibitor, LB-100, combined with atezolizumab, a PD-L1 inhibitor, the proprietary molecule of F. Hoffman-La Roche Ltd. (“Roche”), for patients with microsatellite stable metastatic colorectal cancer. Under the agreement, the Company will provide its lead compound, LB-100, and under a separate agreement between NKI and Roche, Roche will provide atezolizumab and financial support for the clinical trial. The Company has no obligation to and will not provide any reimbursement of clinical trial costs. Pursuant to the agreement and the protocol set forth in the agreement, the clinical trial will be conducted by NKI at NKI’s site in Amsterdam by principal investigator Neeltje Steeghs, MD, PhD, and NKI will be responsible for the recruitment of patients. The agreement provides for the protection of the respective intellectual property rights of each of the Company, NKI and Roche.
This Phase 1b clinical trial will evaluate safety, optimal dose and preliminary efficacy of LB-100 combined with atezolizumab for the treatment of patients with metastatic microsatellite stable colorectal cancer. Immunotherapy using monoclonal antibodies like atezolizumab can enhance the body’s immune response against cancer and hinder tumor growth and spread. LB-100 has been found to improve the effectiveness of anticancer drugs in killing cancer cells by inhibiting a protein called PP2A on cell surfaces. Blocking PP2A increases stress signals in tumor cells expressing the PP2A protein. Accordingly, combining atezolizumab with LB-100 may enhance treatment efficacy for metastatic colorectal cancer, as cancer cells with heightened stress signals are more vulnerable to immunotherapy.
This study comprises a dose escalation phase and a dose expansion phase. The objective of the dose escalation phase is to determine the recommended Phase 2 dose (RP2D) of LB-100 when combined with the standard dosage of atezolizumab. The dose expansion phase will further investigate the preliminary efficacy, safety, tolerability, and pharmacokinetics/dynamics of the LB-100 and atezolizumab combination. The clinical trial opened in August 2024 with the enrollment of the first patient. A total of two patients have been enrolled to date. Patient accrual is expected to take up to 24 months, with a maximum of 37 patients with advanced colorectal cancer to be enrolled in this study.
The principal investigator of the colorectal study testing LB-100 in combination with atezolizumab is currently investigating two Serious Adverse Events (“SAEs”) observed in the clinical trial. The Investigational Review Board (IRB) of NKI has requested additional information with respect to these SAEs and the study has been paused for enrollment until the IRB’s questions have been satisfactorily addressed (see “Specific Risks Associated with the Company’s Business Activities - Serious Adverse Events” below for additional information).
The Company has no financial contractual commitment associated with this clinical trial.
GEIS. Effective July 31, 2019, the Company entered into a Collaboration Agreement for an Investigator-Initiated Clinical Trial with the Spanish Sarcoma Group (Grupo Español de Investigación en Sarcomas or “GEIS”), Madrid, Spain, to carry out a study entitled “Randomized phase I/II trial of LB-100 plus doxorubicin vs. doxorubicin alone in first line of advanced soft tissue sarcoma”. The purpose of this clinical trial is to obtain information with respect to the efficacy and safety of LB-100 combined with doxorubicin in soft tissue sarcomas. Doxorubicin is the global standard for initial treatment of advanced soft tissue sarcomas (“ASTS”). Doxorubicin alone has been the mainstay of first line treatment of ASTS for over 40 years, with little improvement in survival from adding cytotoxic compounds to or substituting other cytotoxic compounds for doxorubicin. In animal models, LB-100 has consistently enhanced the anti-tumor activity of doxorubicin without apparent increases in toxicity.
| 24 |
GEIS has a network of referral centers in Spain and across Europe that have an impressive track record of efficiently conducting innovative studies in ASTS. The Company agreed to provide GEIS with a supply of LB-100 to be utilized in the conduct of this clinical trial, as well as to provide funding for the clinical trial. The goal is to enter approximately 150 to 170 patients in this clinical trial over a period of two to four years. The Phase 1 portion of the study began in the quarter ended June 30, 2023 to determine the recommended Phase 2 dose of the combination of doxorubicin and LB-100. As advanced sarcoma is a very aggressive disease, the design of the Phase 2 portion of the study assumes a median progression-free survival (“PFS”), no evidence of disease progression or death from any cause, of 4.5 months in the doxorubicin arm and an alternative median PFS of 7.5 months in the doxorubicin plus LB-100 arm to demonstrate a statistically significant decrease in relative risk of progression or death by adding LB-100. There is a planned interim analysis of the primary endpoint when approximately 50% of the 102 events required for final analysis is reached.
The Company had previously expected that this clinical trial would commence during the quarter ended June 30, 2020. However, during July 2020, the Spanish regulatory authority advised the Company that although it had approved the scientific and ethical basis of the protocol, it required that the Company manufacture new inventory of LB-100 under current Spanish pharmaceutical manufacturing standards. These standards were adopted subsequent to the production of the Company’s existing LB-100 inventory.
In order to manufacture a new inventory supply of LB-100 for the GEIS clinical trial, the Company engaged a number of vendors to carry out the multiple tasks needed to make and gain approval of a new clinical product for investigational study in Spain. These tasks included the synthesis under good manufacturing practice (GMP) of the active pharmaceutical ingredient (API), with documentation of each of the steps involved by an independent auditor. The API was then transferred to a vendor that prepares the clinical drug product, also under GMP conditions documented by an independent auditor. The clinical drug product was then sent to a vendor to test for purity and sterility, provide appropriate labels, store the drug, and distribute the drug to the clinical centers for use in the clinical trials. A formal application documenting all steps taken to prepare the clinical drug product for clinical use was submitted to the appropriate regulatory authorities for review and approval before being used in a clinical trial.
On October 13, 2022, the Company announced that the Spanish Agency for Medicines and Health Products (Agencia Española de Medicamentos y Productos Sanitarios or “AEMPS”) had authorized a Phase 1b/randomized Phase 2 study of LB-100, the Company’s lead clinical compound, plus doxorubicin, versus doxorubicin alone, the global standard for initial treatment of ASTS. Consequently, this clinical trial commenced during the quarter ended June 30, 2023 and is expected to be completed and a report prepared by December 31, 2026. In April 2023, GEIS completed its first site initiation visit in preparation for the clinical trial at Fundación Jiménez Díaz University Hospital (Madrid). Up to 170 patents will be entered into the clinical trial. The recruitment for the Phase 1b portion of the protocol was extended with two patients and was completed during the quarter ended September 30, 2024. The Company expects to have data on toxicity and preliminary efficacy from this portion of the clinical trial during the quarter ending December 31, 2025.
Given
the focus on the combination of LB-100 with immunotherapy in ovarian clear cell carcinoma and colorectal cancer and the availability
of capital resources, the Company entered into Amendment No. 1 to the Collaboration Agreement effective March 11, 2025 that relieved
the Company of the financial obligation to support the randomized Phase 2 portion of the clinical trial contemplated in the Collaboration
Agreement of approximately $
The Company’s agreement with GEIS provided for various payments based on achieving specific milestones over the term of the agreement. During the three months ended March 31, 2026 and 2025, the Company did not incur any costs pursuant to this agreement.
The
Company’s aggregate commitment pursuant to this agreement, less amounts previously paid to date, totaled approximately $
| 25 |
MD Anderson Cancer Center Clinical Trial. On September 20, 2023, the Company announced an investigator-initiated Phase 1b/2 collaborative clinical trial to assess whether adding LB-100 to a human programmed death receptor-1 (“PD-1”) blocking antibody of GSK plc (“GSK”), dostarlimab-gxly, may enhance the effectiveness of immunotherapy in the treatment of ovarian clear cell carcinoma (“OCCC”). The study objective is to determine the overall survival (“OS”) of patients with OCCC. The clinical trial is being sponsored by The University of Texas MD Anderson Cancer Center (“MD Anderson”) and is being conducted at The University of Texas - MD Anderson Cancer Center. The Company is providing LB-100 and GSK is providing dostarlimab-gxly and financial support for the clinical trial. On January 29, 2024, the Company announced the entry of the first patient into this clinical trial. The Company currently expects that this clinical trial will be completed by December 31, 2027.
On February 25, 2025, the Company announced that it has added the Robert H. Lurie Comprehensive Cancer Center (Lurie Cancer Center) of Northwestern University as a second site in a clinical trial combining the Company’s proprietary compound LB-100 with GSK’s dostarlimab to treat ovarian clear cell cancer. Patient recruitment is underway, and the first patient has been dosed.
Clinical Trial Monitoring Agreements
MD Anderson Cancer Center Clinical Trial. On May 15, 2024, the Company signed a letter of intent with Theradex to monitor the MD Andersen investigator-initiated Phase 1b/2 collaborative clinical trial to assess whether adding LB-100 to a human programmed death receptor-1 (“PD-1”) blocking antibody of GSK plc (“GSK”), dostarlimab-gxly, may enhance the effectiveness of immunotherapy in the treatment of ovarian clear cell carcinoma (“OCCC”). On August 19, 2024, the Company signed a work order agreement with Theradex to monitor the MD Anderson clinical trial. The study oversight is expected to be completed by January 31, 2027.
Costs
under this letter of intent and related work order agreement are estimated to be approximately $
The
Company’s aggregate commitment pursuant to this letter of intent, less amounts previously paid to date, totaled approximately $
City
of Hope. On February 5, 2021, the Company signed a new work order agreement with Theradex to monitor the City of Hope investigator-initiated
clinical trial in small cell lung cancer in accordance with FDA requirements for oversight by the sponsoring party. Costs under this
work order agreement were estimated to be approximately $
GEIS. On June 22, 2023, the Company finalized a work order agreement with Theradex, to monitor the GEIS investigator-initiated clinical Phase I/II randomized trial of LB-100 plus doxorubicin vs. doxorubicin alone in first line of advanced soft tissue sarcoma. The study oversight is expected to be completed by December 31, 2026.
Costs
under this work order agreement are estimated to be approximately $
The
Company’s aggregate commitment pursuant to this clinical trial monitoring agreement, less amounts previously paid to date, totaled
approximately $
Netherlands Cancer Institute. On August 27, 2024, the Company finalized a work order agreement with Theradex, to monitor the NKI Phase 1b clinical trial of LB-100 combined with atezolizumab, a PD-L1 inhibitor, for patients with microsatellite stable metastatic colorectal cancer. The study oversight was expected to be completed by May 31, 2027.
| 26 |
Costs
under this work order agreement were estimated to be approximately $
The
Company’s aggregate commitment pursuant to this clinical trial monitoring agreement, less amounts previously paid to date, totaled
approximately $
Patent and License Agreements
National Institute of Health. Effective February 23, 2024, the Company entered into a Patent License Agreement (the “License Agreement”) with the National Institute of Neurological Disorders and Stroke (“NINDS”) and the National Cancer Institute (“NCI”), each an institute or center of the National Institute of Health (“NIH”). Pursuant to the License Agreement, the Company has licensed on an exclusive basis the NIH’s intellectual property rights claimed for a Cooperative Research and Development Agreement (“CRADA”) subject invention co-developed with the Company, and the licensed field of use, which focuses on promoting anti-cancer activity alone, or in combination with standard anti-cancer drugs. The scope of this clinical research extends to checkpoint inhibitors, immunotherapy, and radiation for the treatment of cancer. The License Agreement is effective, and shall extend, on a licensed product, licensed process, and country basis, until the expiration of the last-to-expire valid claim of the jointly owned licensed patent rights in each such country in the licensed territory, estimated at twenty years, unless sooner terminated.
The License Agreement contemplates that the Company will seek to work with pharmaceutical companies and clinical trial sites (including comprehensive cancer centers) to initiate clinical trials within timeframes that will meet certain benchmarks. Data from the clinical trials will be the subject of various regulatory filings for marketing approval in applicable countries in the licensed territories. Subject to the receipt of marketing approval, the Company would be expected to commercialize the licensed products in markets where regulatory approval has been obtained.
The
Company is obligated to pay the NIH an annual royalty of $
The Company is obligated to pay the NIH, on a country-by-country basis, earned royalties of 2% on net sales of each royalty-bearing product and process, subject to reduction by 50% under certain circumstances relating to royalties paid by the Company to third parties, but not less than 1%. The Company’s obligation to pay earned royalties under the License Agreement commences on the date of the first commercial sale of a royalty-bearing product or process and expires on the date on which the last valid claim of the licensed product or licensed process expires in such country.
The
Company is obligated to pay the NIH benchmark royalties, on a one-time basis, within sixty days from the first achievement of each such
benchmark. The License Agreement defines four such benchmarks, which the Company is required to pursue based on “commercially reasonable
efforts” as defined in the License Agreement, with deadlines of October 1, 2024, 2027, 2029 and 2031, each with a different specified
benchmark payment amount payable within thirty days of achieving such benchmark. The October 1, 2024 benchmark of $
The Company is obligated to provide annual reports to the NIH on its progress toward the development and commercialization of products under the licensed patents. These reports, due within sixty days following the end of each calendar year, must include updates on research and development activities, regulatory submissions, manufacturing efforts, sublicensing, and sales initiatives. If any deviations from the established commercial development plan or agreed-upon benchmarks occur, the Company is obligated to provide explanation and may amend the commercial development plan and the benchmarks, which, subject to certain conditions, the NIH shall not unreasonably withhold, condition, or delay approval of any request of the Company to amend the commercial development plan and/or the benchmarks and to extend the time periods of the benchmarks.
| 27 |
The
Company is obligated to pay the NIH sublicensing royalties of
During
the three months ended March 31, 2026 and 2025, the Company incurred costs of $
The
Company’s aggregate commitment pursuant to this agreement, less amounts previously paid to date, totaled approximately $
Other Significant Agreements and Contracts
BioPharmaWorks. Effective September 14, 2015, the Company entered into a Collaboration Agreement with BioPharmaWorks, pursuant to which the Company engaged BioPharmaWorks to perform certain services for the Company.
The
Company recorded charges to operations pursuant to this Collaboration Agreement of $
Netherlands Cancer Institute. On October 8, 2021, the Company entered into a Development Collaboration Agreement with the Netherlands Cancer Institute, Amsterdam (“NKI”) (see Note 5), one of the world’s leading comprehensive cancer centers, and Oncode Institute, Utrecht, a major independent cancer research center, for a term of three years. The Development Collaboration Agreement was subsequently modified by Amendment No. 1 thereto.
The
Development Collaboration Agreement is a preclinical study intended to identify the most promising drugs to be combined with LB-100,
and potentially LB-100 analogues, to be used to treat a range of cancers, as well as to identify the specific molecular mechanisms underlying
the identified combinations. The Company agreed to fund the preclinical study, at an approximate cost of
On
October 3, 2023, the Company entered into Amendment No. 2 to the Development Collaboration Agreement with NKI, which provides for additional
research activities, extends the termination date of the Development Collaboration Agreement by two years to October 8, 2026, and added
| 28 |
On
October 4, 2024, the Company entered into Amendment No. 3 to the Development Collaboration Agreement with NKI, which suspended Amendment
No. 2 and provided for a new study term of one year commencing upon the dosing of the first patient in the trial at a project cost of
During
the three months ended March 31, 2026 and 2025, the Company incurred charges of $
MRI
Global. As amended, the Company has contracted with MRI Global for stability analysis, storage and distribution of LB-100 for clinical
trials in the United States. During the three months ended March 31, 2026 and 2025, the Company incurred costs of $
Specific Risks Associated with the Company’s Business Activities
Serious Adverse Events
The Company’s lead drug candidate, LB-100, is currently undergoing various clinical trials, and there is a risk that one or more of these trials could be placed on hold by regulatory authorities due to serious adverse events (SAEs) related to the Company’s drug candidate or to another company’s drug used in combination in one of the Company’s clinical trials. It is possible that the SAEs could be attributable to the Company’s drug candidate and could include, but not be limited to, unexpected severe side effects, treatment-related deaths, or long-term health complications. A dose given could result in non-tolerable adverse events defined as dose-limiting toxicity (DLT). When two DLTs occur at the same dose-level that dose-level is considered too high and unsafe. Further treatment is only allowed at lower dose-levels that have previously been found safe.
If an SAE or a pattern of SAEs is observed during the course of a clinical trial involving the Company’s drug candidate, the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), or other regulatory authorities may issue a clinical hold, requiring the Company to pause or discontinue further enrollment and dosing in the Company’s clinical trial. It is also possible that the clinical trial could be terminated. Any of these actions could delay or halt the development of the Company’s drug candidate, increase development costs, and negatively impact the Company’s ability to ultimately achieve regulatory approval. Additionally, if an SAE is confirmed to be drug-related, the Company may be required to conduct additional studies, modify the study design, or abandon further development of the drug candidate altogether, which could materially impact the Company’s business, financial condition, and prospects.
The occurrence of an SAE and any resulting clinical hold could also harm the Company’s reputation with patients, physicians, health institutions, and investors, diminish the Company’s ability to attract clinical trial participants, and damage the Company’s ability to interest investors and obtain financing in the future. There can be no assurances that the Company will not experience such SAEs in the future or that any related clinical hold will be lifted in a timely manner, or at all.
The principal investigator of the colorectal study testing LB-100 in combination with atezolizumab (Roche PD-L1 inhibitor) is currently investigating two SAEs observed in the clinical trial that was launched in August 2024. The Institutional Review Board (the “IRB”) of the Netherlands Cancer Institute (“NKI”) has put the colorectal cancer study on hold. The adverse reactions that developed in the two patients were dyspnea (shortness of breath) due to lung toxicity possibly or probably related to the combination of LB-100 and atezolizumab in one patient and fever and aphasia possibly or probably related to the combination of LB-100 and atezolizumab in the second patient. The patient who developed lung toxicity deceased due to the combination of lung metastases of colorectal cancer and dyspnea. The patient with fever and aphasia fully recovered from the adverse events with supportive medication.
Given the identified adverse events in the two patients in the clinical trial, the IRB requested from the principal investigator of the study at the NKI information as to whether the adverse events could have been caused by the combination of LB-100 and atezolizumab and information about the mode of action of the combination of LB-100 and atezolizumab. The principal investigator prepared a response to the IRB detailing the safety experience with LB-100 given alone and in combination with other cancer drugs, especially doxorubicin and dostarlimab. Doxorubicin is a well-known chemotherapy, and dostarlimab is a well-known immunotherapy of which the mode of action is closely related to that of atezolizumab.
The reported adverse events in the colorectal cancer study have not been seen in any other patients thus far treated with LB-100 alone or in combination with other cancer drugs. Through September 30,2025, the Company has been informed that a total of 86 patients had received or were receiving experimental treatment with LB-100.
In May 2025, the Company updated the safety overview of LB-100 and delivered the updated version 5.0 of the Investigator’s Brochure (the “IB”), which contains all of the relevant preclinical, clinical and pharmacologic data with respect to the study of the LB-100 clinical compound in humans, to the investigators of all ongoing clinical trials. The investigators of the study in colorectal cancer (NCT06012734) submitted a detailed response to the IRB, including the updated IB. The Company is currently awaiting the outcome of the IRB review.
| 29 |
Other Business Risks
The Company is also subject to additional risks and uncertainties arising from changes to the macroeconomic environment and geopolitical events. U.S. and global financial markets have experienced volatility and disruption due to macroeconomic and geopolitical events such as the implementation of tariffs, inflation, the risk of a recession and ongoing conflicts in other countries. In addition, if equity and credit markets deteriorate, it may make any future debt or equity financing more difficult to obtain on favorable terms, and potentially more dilutive to existing stockholders. The Company cannot predict at this time to what extent it and its collaborators, employees, suppliers, contract manufacturers and/or vendors could potentially be negatively impacted by these events.
The Company has established policies and processes for assessing, identifying and managing material risk from cybersecurity threats, and has integrated these processes into its overall risk management systems and processes. The Company routinely assesses material risks from cybersecurity threats, including any potential unauthorized occurrence on or conducted through its information and email systems that may result in adverse effects on the confidentiality, integrity, or availability of the Company’s information and email systems or any information residing therein. The Company conducts periodic risk assessments to identify cybersecurity threats, as well as assessments in the event of a material change in the Company’s business practices that may affect information systems that are vulnerable to such cybersecurity threats. These risk assessments include identification of reasonably foreseeable internal and external risks, the likelihood and potential damage that could result from such risks, and the sufficiency of existing policies, procedures, systems and safeguards in place to manage such risks. The Company has not encountered any cybersecurity challenges to date that have materially impaired its operations or financial condition.
10. Subsequent Events
On April 15, 2026, the Compensation Committee of the Board of Directors approved the cancellation of certain outstanding stock options previously granted to the Company’s current officers and directors and, in replacement thereof, granted restricted share units (“RSUs”) under the Company’s 2020 Stock Incentive Plan. The Company will recognize an incremental cost if the fair value is increased as a result of the modification.
On May 5, 2026,
| 30 |
ITEM 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Forward-Looking Statements
This Quarterly Report on Form 10-Q of Lixte Biotechnology Holdings, Inc. (the “Company”) contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, and Section 21E of the Securities Exchange Act of 1934. These might include statements regarding the Company’s financial position, business strategy and other plans and objectives for future operations, and assumptions and predictions about future clinical trials and their timing and costs, product demand, supply, manufacturing costs, marketing and pricing factors are all forward-looking statements. These statements are generally accompanied by words such as “intend”, “anticipate”, “believe”, “estimate”, “potential(ly)”, “continue”, “forecast”, “predict”, “plan”, “may”, “will”, “could”, “would”, “should”, “expect” or the negative of such terms or other comparable terminology. The Company believes that the assumptions and expectations reflected in such forward-looking statements are reasonable, based on information available to it on the date hereof, but the Company cannot provide assurances that these assumptions and expectations will prove to have been correct or that the Company will take any action that the Company may presently be planning. These forward-looking statements are inherently subject to known and unknown risks and uncertainties. Actual results or experience may differ materially from those expected, anticipated or implied in the forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to, regulatory policies or changes thereto, available cash, research and development results, competition from other similar businesses, and market and general economic factors. This discussion should be read in conjunction with the condensed consolidated financial statements and notes thereto included in Item 1 of this Quarterly Report on Form 10-Q and the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2024, including the section entitled “Item 1A. Risk Factors”. The Company does not intend to update or revise any forward-looking statements to reflect new information, future events or otherwise.
Overview
The Company is a clinical-stage biopharmaceutical company focused on identifying new targets for cancer drug development and developing and commercializing cancer therapies. The Company’s corporate office is located in Pasadena, California.
The Company’s product pipeline is primarily focused on inhibitors of protein phosphatase 2A, which is used to enhance cytotoxic agents, radiation, immune checkpoint blockers and other cancer therapies. The Company believes that inhibitors of protein phosphatases have significant therapeutic potential for a broad range of cancers. The Company is focusing on the clinical development of a specific protein phosphatase inhibitor, referred to as LB-100.
The Company’s activities are subject to significant risks and uncertainties, including the need for additional capital. The Company has not yet commenced any revenue-generating operations, does not have positive cash flows from operations, relies on stock-based compensation for a substantial portion of employee and consultant compensation, and is dependent on periodic access to equity capital to fund its operating requirements.
Going Concern
For the three months ended March 31, 2026, the Company incurred a net loss of $1,987,608 and used cash in operations of $1,831,363. As of March 31, 2026, the Company had cash of $3,250,650 available to fund its operations. The Company has not generated recurring revenues since inception and has incurred negative operating cash flows as it advances its development programs. As a result, management has concluded that there is a substantial doubt regarding the Company’s ability to continue as a going concern for a period of at least 12 months beyond the filing of this quarterly report. In addition, our independent registered public accounting firm, in their audit report to the financial statements included in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025, expressed substantial doubt about the Company’s ability to continue as a going concern. The condensed consolidated financial statements have been prepared assuming the Company will continue as a going concern and do not include any adjustments that might result from the outcome of this uncertainty.
The Company is currently engaged in early-stage clinical trials for its lead product candidate, LB-100. These activities require substantial research, development, regulatory, and clinical expenditures, and the Company does not expect to generate sustainable operating revenues for several years, if ever. At March 31, 2026, the Company’s remaining contractual commitments pursuant to clinical trial agreements and clinical trial monitoring agreements aggregated approximately $482,702 which are expected to be incurred through December 31, 2027.
In addition, through the acquisition of Liora, the Company expects to incur approximately $2 million over the next 24 months to recommission and update the Light machine. Liora currently has no revenues, and the Company will require additional capital to fund these activities.
| 31 |
Management is actively evaluating and pursuing additional financing alternatives, including equity and debt financing and potential strategic transactions. However, there can be no assurance that additional funding will be available on acceptable terms, in sufficient amounts, or at all. If the Company is unable to obtain the necessary funding, it may be required to delay, scale back, or eliminate its clinical development programs; curtail expenditures related to the LiGHT system; or pursue strategic alternatives, including potential asset sales or the cessation of operations.
Nasdaq Compliance
The Company’s common stock are traded on the Nasdaq Capital Market under the symbol “LIXT”.
On August 23, 2024, the Company received written notification from the Listing Qualifications Department (the “Staff”) of the Nasdaq Stock Market LLC (“Nasdaq”) that the Company was not in compliance with the minimum stockholders’ equity requirement of $2,500,000 for continued listing on the Nasdaq Capital Market under Listing Rule 5550(b) (the “Stockholders’ Equity Requirement”). On October 3, 2024, the Company submitted a plan to the Staff to regain compliance with the Stockholders’ Equity Requirement. On October 21, 2024, the Staff provided written notification to the Company that it had granted an extension through February 18, 2025 to regain compliance with the Stockholders’ Equity Requirement. As of February 18, 2025, the Company had not gained compliance with the Stockholders’ Equity Requirement. Accordingly, on February 19, 2025, the Company received written notification from the Staff stating that the Company did not meet the terms of the extension because it did not complete its proposed financing initiatives to regain compliance. The Company timely requested a hearing before the Nasdaq Hearings Panel (“Panel”), staying any suspension or delisting pending the Panel’s decision. Following an April 3, 2025 hearing, the Panel granted the Company a further extension through July 3, 2025 to regain compliance. On July 2, 2025, the Company closed a $5.05 million private placement and, on July 8, 2025, completed a $1.5 million registered direct offering. On July 15, 2025, Nasdaq notified the Company that it had regained compliance with the stockholders’ equity requirement.
The Company remains subject to a Panel Monitor under Nasdaq Listing Rule 5815(d)(4)(B) through July 15, 2026. During this period, any future deficiency in stockholders’ equity would require the Company to request a hearing before the Panel rather than submit a new compliance plan.
Recent Accounting Pronouncements
Information with respect to recent accounting pronouncements is provided at Note 2 to the condensed consolidated financial statements for the three months ended March 31, 2026 and 2025 included elsewhere in this document.
Critical Accounting Policies and Estimates
The Company’s significant accounting policies are described in Note 2 to the condensed consolidated financial statements for the three months ended March 31, 2026 and 2025 included elsewhere in this document. These policies, together with the related estimates and assumptions, are fundamental to understanding the Company’s results of operations and financial condition. Management evaluates these policies and estimates on an ongoing basis based on historical experience, current conditions, and other factors deemed relevant.
There have been no material changes to the Company’s critical accounting policies and estimates from those disclosed in the Company’s most recent Annual Report on Form 10-K.
| 32 |
Summary of Business Activities and Plans
Company Overview
The Company is a clinical-stage biopharmaceutical company focused on identifying new targets for cancer drug development and developing and commercializing cancer therapies. The Company’s product pipeline is primarily focused on inhibitors of protein phosphatase 2A, which is used to enhance cytotoxic agents, radiation, immune checkpoint blockers and other cancer therapies. The Company believes that inhibitors of protein phosphatases have significant therapeutic potential for a broad range of cancers. The Company is focusing on the clinical development of a specific protein phosphatase inhibitor, referred to as LB-100.
The Company believes that the mechanism by which LB-100 affects cancer cell growth is different from cancer agents currently approved for clinical use. LB-100 is currently being tested in clinical trials in Ovarian Clear Cell Carcinoma, Metastatic Colon Cancer, and Advanced Soft Tissue Sarcoma. LB-100 has shown anti-cancer activity in animal models of glioblastoma multiforme, neuroblastoma, and medulloblastoma, all cancers of neural tissue. LB-100 has also been shown to enhance the effectiveness of commonly used anti-cancer drugs in animal models of melanoma, breast cancer and sarcoma. The enhancement of anti-cancer activity of these anti-cancer drugs occurs at doses of LB-100 that do not significantly increase toxicity in animals. It is therefore hoped that, when combined with standard anti-cancer regimens against many tumor types, LB-100 will improve therapeutic benefit.
As a compound moves through the FDA-approval process, it becomes an increasingly valuable property, but at a cost of additional investment at each stage. As the potential effectiveness of LB-100 has been documented at the clinical trial level, the Company has allocated resources to manage its patent portfolio. The Company’s approach has been to operate with a minimum of overhead, moving compounds forward as efficiently and inexpensively as possible, and to raise funds to support each of these stages as certain milestones are reached. The Company’s longer-term objective is to secure one or more strategic partnerships or licensing agreements with pharmaceutical companies with major programs in cancer.
Specific Risks Associated with the Company’s Business Activities
Serious Adverse Events
The Company’s lead drug candidate, LB-100, is currently undergoing various clinical trials, and there is a risk that one or more of these trials could be placed on hold by regulatory authorities due to serious adverse events (SAEs) related to the Company’s drug candidate or to another company’s drug used in combination in one of the Company’s clinical trials. It is possible that the SAEs could be attributable to the Company’s drug candidate and could include, but not be limited to, unexpected severe side effects, treatment-related deaths, or long-term health complications. A dose given could result in non-tolerable adverse events defined as dose-limiting toxicity (DLT). When two DLTs occur at the same dose-level that dose-level is considered too high and unsafe. Further treatment is only allowed at lower dose-levels that have previously been found safe.
If an SAE or a pattern of SAEs is observed during the course of a clinical trial involving the Company’s drug candidate, the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), or other regulatory authorities may issue a clinical hold, requiring the Company to pause or discontinue further enrollment and dosing in the Company’s clinical trial. It is also possible that the clinical trial could be terminated. Any of these actions could delay or halt the development of the Company’s drug candidate, increase development costs, and negatively impact the Company’s ability to ultimately achieve regulatory approval. Additionally, if an SAE is confirmed to be drug-related, the Company may be required to conduct additional studies, modify the study design, or abandon further development of the drug candidate altogether, which could materially impact the Company’s business, financial condition, and prospects.
The occurrence of an SAE and any resulting clinical hold could also harm the Company’s reputation with patients, physicians, health institutions, and investors, diminish the Company’s ability to attract clinical trial participants, and damage the Company’s ability to interest investors and obtain financing in the future. There can be no assurances that the Company will not experience such SAEs in the future or that any related clinical hold will be lifted in a timely manner, or at all.
| 33 |
The principal investigator of the colorectal study testing LB-100 in combination with atezolizumab (Roche PD-L1 inhibitor) is currently investigating two SAEs observed in the clinical trial that was launched in August 2024. The Institutional Review Board (the “IRB”) of the Netherlands Cancer Institute (“NKI”) has put the colorectal cancer study on hold. The adverse reactions that developed in the two patients were dyspnea (shortness of breath) due to lung toxicity possibly or probably related to the combination of LB-100 and atezolizumab in one patient and fever and aphasia possibly or probably related to the combination of LB-100 and atezolizumab in the second patient. The patient who developed lung toxicity deceased due to the combination of lung metastases of colorectal cancer and dyspnea. The patient with fever and aphasia fully recovered from the adverse events with supportive medication.
Given the identified adverse events in the two patients in the clinical trial, the IRB requested from the principal investigator of the study at the NKI information as to whether the adverse events could have been caused by the combination of LB-100 and atezolizumab and information about the mode of action of the combination of LB-100 and atezolizumab. The principal investigator prepared a response to the IRB detailing the safety experience with LB-100 given alone and in combination with other cancer drugs, especially doxorubicin and dostarlimab. Doxorubicin is a well-known chemotherapy, and dostarlimab is a well-known immunotherapy of which the mode of action is closely related to that of atezolizumab.
The reported adverse events in the colorectal cancer study have not been seen in any other patients thus far treated with LB-100 alone or in combination with other cancer drugs. Through early July 2025, the Company has been informed that a total of 82 patients had received or were receiving experimental treatment with LB-100.
In May 2025, the Company updated the safety overview of LB-100 and delivered the updated version 5.0 of the Investigator’s Brochure (the “IB”), which contains all of the relevant preclinical, clinical and pharmacologic data with respect to the study of the LB-100 clinical compound in humans, to the investigators of all ongoing clinical trials. The investigators of the study in colorectal cancer (NCT06012734) submitted a detailed response to the IRB, including the updated IB. The Company is currently awaiting the outcome of the IRB review.
External Risks Associated with the Company’s Business Activities
Covid-19 Virus. The global outbreak of the novel coronavirus (Covid-19) in early 2020 led to disruptions in general economic activities throughout the world as businesses and governments implemented broad actions to mitigate this public health crisis. Although Covid-19 outbreak has subsided, the extent to which the coronavirus pandemic may reappear and impact the Company’s clinical trial programs and capital raising efforts in the future is uncertain and cannot be predicted.
Inflation and Interest Rate Risk. The Company does not believe that inflation or increasing interest rates have had a material effect on its operations to date, other than their impact on the general economy. However, there is a risk that the Company’s operating costs could become subject to inflationary and interest rate pressures in the future, which would have the effect of increasing the Company’s operating costs, and which would put additional stress on the Company’s working capital resources.
Supply Chain Issues. The Company does not currently expect that supply chain issues will have a significant impact on its business activities, including its ongoing clinical trials.
Potential Recession. There have been some indications that the United States economy may be at risk of entering a recessionary period. Although it does not appear likely at this time, an economic recession could impact the general business environment and the capital markets, which could, in turn, affect the Company.
Geopolitical Risk. The geopolitical landscape poses inherent risks that could significantly impact the operations and financial performance of the Company. In the event of a military conflict, supply chain disruptions, geopolitical uncertainties, and economic repercussions may adversely affect the Company’s ability to conduct research, develop, test and manufacture products, and distribute them globally. This could lead to delays in product development, interruptions in the supply of critical materials, and delays in clinical trials, thereby impeding the Company’s clinical development and commercialization plans. Furthermore, the impact of a conflict on global financial markets may result in increased volatility and uncertainty in the capital markets, thereby affecting the valuation of the Company’s publicly-traded shares. Investor confidence, market sentiment, and access to capital could all be negatively influenced. Such geopolitical risks are outside the control of the Company, and the actual effects on the Company’s business, financial condition and results of operations may differ from current estimates.
| 34 |
Cybersecurity Risks. The Company has established policies and processes for assessing, identifying and managing material risk from cybersecurity threats, and has integrated these processes into its overall risk management systems and processes. The Company routinely assesses material risks from cybersecurity threats, including any potential unauthorized occurrence on or conducted through its information and email systems that may result in adverse effects on the confidentiality, integrity, or availability of the Company’s information and email systems or any information residing therein. The Company conducts periodic risk assessments to identify cybersecurity threats, as well as assessments in the event of a material change in the Company’s business practices that may affect information systems that are vulnerable to such cybersecurity threats. These risk assessments include identification of reasonably foreseeable internal and external risks, the likelihood and potential damage that could result from such risks, and the sufficiency of existing policies, procedures, systems and safeguards in place to manage such risks. The Company has not encountered any cybersecurity challenges to date that have materially impaired its operations or financial condition.
The Company is continuing to monitor these matters and will adjust its current business and financing plans as more information becomes available.
Results of Operations
At March 31, 2026, the Company had not yet commenced any revenue-generating operations, does not have any positive cash flows from operations, and is dependent on its ability to raise equity capital to fund its operating requirements.
The Company’s condensed consolidated statements of operations as discussed herein are presented below.
| Three Months Ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Revenues, net | $ | - | $ | - | ||||
| Costs and expenses: | ||||||||
| General and administrative costs | 1,654,031 | 615,483 | ||||||
| Research and development costs | 333,376 | 91,457 | ||||||
| Total costs and expenses | 1,987,407 | 706,940 | ||||||
| Loss from operations | (1,987,407 | ) | (706,940 | ) | ||||
| Other income (expenses): | ||||||||
| Interest Income | 3,370 | 441 | ||||||
| Interest Expense | (3,094 | ) | (3,135 | ) | ||||
| Foreign currency gain (loss) | (475 | ) | 79 | |||||
| Net loss | $ | (1,987,608 | ) | $ | (709,555 | ) | ||
| Series B Convertible Preferred Stock 8% cumulative dividend | (17,882 | ) | - | |||||
| Non-controlling interest | 63,932 | - | ||||||
| Net loss attributable to common stockholders | (1,941,558 | ) | (709,555 | ) | ||||
| Net loss per common share – basic and diluted | $ | (0.18 | ) | $ | (0.29 | ) | ||
| Weighted average common shares outstanding – basic and diluted | 10,889,003 | 2,471,513 | ||||||
| 35 |
Three Months Ended March 31, 2026 and 2025
Revenues. The Company did not have any revenues for the three months ended March 31, 2026 and 2025.
Research and Development Costs. For the three months ended March 31, 2026, research and development costs were $333,376 which consisted of clinical and related oversight costs of $309,794 and compound maintenance costs of $5,516, and preclinical research focused on development of additional novel anti-cancer compounds to add to the Company’s clinical pipeline of $13,566.
For the three months ended March 31, 2025, research and development costs were $91,457, which consisted of clinical and related oversight costs of $15,868, compound maintenance costs of $32,819, and preclinical research focused on development of additional novel anti-cancer compounds to add to the Company’s clinical pipeline of $42,770.
Research and development expenses increased by $241,919, or 265%, in 2026 as compared to 2025. The increase was primarily attributable to a final payment related to clinical trial engagement which increased the cost by $293,297, partially offset by a decrease in compound maintenance costs of $56,507.
General and Administrative Costs. For the three months ended March 31, 2026, general and administrative costs were $1,654,031 which primarily consisted of Accounting fees of $413,504, Investment banking fees of $250,000, payroll expenses of $217,927, public relations fees of $178,500, consulting fees related to Liora acquisition of $172,572, legal fees of $37,590, lease expense of $145, 365, Insurance expense including D&O of $64,387, intellectual property costs of $35,032, listing fees of $14,000, Investor relations of $13,797, licenses and royalties of $7,500, and stock-based compensation to related parties of $82,176.
For the three months ended March 31, 2025, general and administrative costs were $615,483, which consisted of the fair value of vested stock options issued to directors and officers of $99,738 (including quarterly director and board committee fees of $27,500), patent and licensing legal and filing fees and costs of $56,084, other consulting and professional fees of $205,315, insurance expense of $64,277, officer compensation and related costs of $118,268, cash-based director and board committee fees of $0, licensing and royalties of $7,397, shareholder reporting costs of $4,811, listing fees of $33,250, filing fees of $9,750, investor relations of $11,397, taxes and licenses of $5,056, and other operating costs of $2,998, offset by a rent refund of $2,858.
| 36 |
General and administrative costs increased by $1,038,548 or 169%, in 2026 as compared to 2025, primarily as a result of increase in accounting fees by $318,485, increase in investment banking fees by $221,140, increase in public relations costs by 178,500, consulting fees related to acquisition by $172,572, increase in lease expense by $145,365, increase in payroll expense by $127,162 partially offset by decrease in legal fees by $43,847, decrease in consulting fees supporting prior period activities with no equivalent services requested in current period, decrease in intellectual property by $21,052.
Interest Income. For the three months ended March 31, 2026, the Company had interest income of $3,370, as compared to interest income of $441 for the three months ended March 31, 2025, due to higher average cash balance and from prevailing higher interest rates compared to three months ended March 31, 2025.
Interest Expense. For the three months ended March 31, 2026, the Company had interest expense of $3,094, as compared to interest expense of $3,135 for the three months ended March 31, 2025, related to the financing of the premium for the Company’s directors and officers liability insurance policy.
Realized Gain (Loss) on Foreign Currency Transactions. For the three months ended March 31, 2026, the Company had a realized foreign currency loss of $475, as compared to a foreign currency gain of $79 for the three months ended March 31, 2025, from foreign currency transactions.
Net Loss. For the three months ended March 31, 2026, the Company incurred a net loss of $1,987,608, as compared to a net loss of $709,555 for the three months ended March 31, 2025.
Liquidity and Capital Resources – March 31, 2026
The Company’s condensed consolidated statements of cash flows as discussed herein are as follows:
| Three Months Ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Net cash used in operating activities | $ | (1,831,363 | ) | $ | (568,483 | ) | ||
| Net cash used in investing activities | (24,859 | ) | — | |||||
| Net cash provided by financing activities | — | 914,228 | ||||||
| Net increase (decrease) in cash | $ | (1,856,222 | ) | $ | 345,745 | |||
At March 31, 2026, the Company had working capital of $2,182,816, as compared to working capital of $3,845,268 at December 31, 2025, reflecting a net decrease in working capital of $1,662,452 for the three months ended March 31, 2026. The decrease in working capital during the three months ended March 31, 2026 was primarily due to operating activities. As of March 31, 2026, the Company had cash of $3,250,650 available to fund its operations.
Going Concern
For the three months ended March 31, 2026, the Company incurred a net loss of $1,987,608 and used cash in operations of $1,831,363. As of March 31, 2026, the Company had cash of $3,250,650 available to fund its operations. The Company has not generated recurring revenues since inception and has incurred negative operating cash flows as it advances its development programs. As a result, management has concluded that there is a substantial doubt regarding the Company’s ability to continue as a going concern for a period of at least 12 months beyond the filing of this quarterly report. In addition, our independent registered public accounting firm, in their audit report to the financial statements included in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025, expressed substantial doubt about the Company’s ability to continue as a going concern.
Consideration of Strategic Alternatives
The Company is focusing on a disciplined approach to strategic expansion and is focused on advancing LB-100 in high-need cancer indications, while pursuing acquisitions of complementary oncology assets that could enhance the Company’s pipeline, accelerate development and create durable value for patients and shareholders. The Company has announced that it is in advanced negotiations regarding potential transactions consistent with its strategy, although there can be no assurance that any transaction will be completed.
The Company will continue to evaluate various alternatives to be able to obtain the capital required to fund its operations and business development activities, and to maintain its listing on the Nasdaq Capital Market, including merger or acquisition opportunities (including reverse mergers and acquisitions) and funding transactions which could result in a change in control of the Company. There can be no assurances that the evaluation process will result in the identification of an appropriate transaction, the negotiation and execution of a definitive agreement to effect such a transaction, or that any such transaction will ultimately be approved by the Company’s stockholders and then be consummated. Even if such a strategic transaction is consummated, there can be no assurances that it would enhance stockholder value, and it may result in substantial dilution to existing stockholders. Any potential transaction would be dependent on a number of factors that may be outside of the control of the Company, including, among other things, market conditions, industry trends, the interest of third parties in a potential transaction with the Company, and the availability of appropriate financing for such a transaction.
Trends, Events and Uncertainties
Research and development of new pharmaceutical compounds by its nature is unpredictable. Although the Company undertakes research and development efforts with commercially reasonable diligence, there can be no assurance that the Company’s cash position will be sufficient to enable it to develop any pharmaceutical compound to the extent needed to create future sales to sustain operations as contemplated herein.
| 37 |
There can be no assurance that the Company’s pharmaceutical compound will obtain the regulatory approvals and market acceptance to achieve sustainable revenues sufficient to support the Company’s operations. Even if the Company is able to generate revenues, there can be no assurance that the Company will be able to achieve operating profitability or positive operating cash flows. There can be no assurance that the Company will be able to secure additional financing, to the extent required, on acceptable terms or at all. If cash resources are insufficient to satisfy the Company’s ongoing cash requirements, the Company would be required to reduce or discontinue its research and development programs, or attempt to obtain funds, if available, through strategic alliances, joint ventures or other transaction structures that could require the Company to relinquish rights to and/or control of LB-100, or to discontinue operations entirely.
Other than as discussed above, the Company is not currently aware of any trends, events or uncertainties that are likely to have a material effect on its financial condition in the near term, although it is possible that new trends or events may develop in the future that could have a material effect on the Company’s financial condition.
ITEM 3. QUANTITATIVE AND QUALITATIVE DISCLOSURE ABOUT MARKET RISK
Not applicable.
ITEM 4. CONTROLS AND PROCEDURES
Disclosure Controls and Procedures
The Company’s management is responsible for establishing and maintaining a system of disclosure controls and procedures (as defined in Rule 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”)), that is designed to ensure that information required to be disclosed by the Company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized, and reported, within the time periods specified in the rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by an issuer in the reports that it files or submits under the Exchange Act is accumulated and communicated to the issuer’s management, including its principal executive officer and principal financial officer, or persons performing similar functions, as appropriate, to allow timely decisions regarding required disclosure.
In accordance with Exchange Act Rules 13a-15 and 15d-15, an evaluation was completed under the supervision and with the participation of the Company’s management, including its Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of the Company’s disclosure controls and procedures as of March 31, 2026, the end of the most recent fiscal period covered by this report. Based on that evaluation, the Company’s management has concluded that the Company’s disclosure controls and procedures were effective in providing reasonable assurance that information required to be disclosed in the Company’s reports filed or submitted under the Exchange Act was recorded, processed, summarized, and reported within the time periods specified in the rules and forms of the Securities and Exchange Commission.
Limitations on Effectiveness of Disclosure Controls and Procedures
In designing and evaluating disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, cannot provide absolute assurance that the objectives of the controls system are met, and no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, within a company have been detected. In addition, the design of disclosure controls and procedures must reflect that there are resource constraints and that management is required to apply judgment in evaluating the benefits of possible controls and procedures relative to their costs.
Changes in Internal Control Over Financial Reporting
The Company’s management, including its Chief Executive Officer and Chief Financial Officer, has determined that no change in the Company’s internal control over financial reporting (as that term is defined in Rules 13(a)-15(f) and 15(d)-15(f) of the Securities Exchange Act of 1934) occurred during the period ended March 31, 2026 that has materially affected, or is reasonably likely to materially affect, the Company’s internal control over financial reporting.
| 38 |
PART II - OTHER INFORMATION
ITEM 1. LEGAL PROCEEDINGS
The Company is not currently subject to any pending or threatened legal actions or claims.
ITEM 1A. RISK FACTORS
The Company’s business, financial condition, results of operations and cash flows may be impacted by a number of factors, many of which are beyond the Company’s control, including those set forth in the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2025, as filed with the Securities and Exchange Commission on March 31, 2026 (the “2025 Form 10-K”).
The Risk Factors set forth in the 2025 Form 10-K should be read carefully in connection with evaluating the Company’s business and in connection with the forward-looking statements contained in this Quarterly Report on Form 10-Q. Any of the risks described in the 2025 Form 10-K could materially adversely affect the Company’s business, financial condition or future results, and the actual outcome of matters as to which forward-looking statements are made. These are not the only risks that the Company faces. Additional risks and uncertainties not currently known to the Company or that the Company currently deems to be immaterial also may materially adversely affect the Company’s business, financial condition and/or operating results.
As of the date of the filing of this document, except as disclosed elsewhere in this document, including Note 9. Subsequent Events, there have been no material changes to the Risk Factors previously disclosed in the Company’s 2025 Form 10-K.
ITEM 2. UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDS
None.
ITEM 3. DEFAULTS UPON SENIOR SECURITIES
Not applicable.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
ITEM 5. OTHER INFORMATION
During
the three months ended March 31, 2026, no director or officer (as defined in Rule 16a-1(f) under the Exchange Act) of the Company
| 39 |
ITEM 6. EXHIBITS
The following documents are filed as part of this report:
* Filed herewith.
| 40 |
SIGNATURES
In accordance with the requirements of the Securities and Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| LIXTE BIOTECHNOLOGY HOLDINGS, INC. | ||
| (Registrant) | ||
| Date: May 14, 2026 | By: | /s/ GEORDAN PURSGLOVE |
| Geordan Pursglove | ||
| President and Chief Executive Officer | ||
| (Principal Executive Officer) | ||
| Date: May 14, 2026 | By: | /s/ PETER STAZZONE |
| Peter Stazzone | ||
| Chief Financial Officer | ||
| (Principal Financial and Accounting Officer) | ||
| 41 |